For over 40 years, Butterworth Laboratories has provided independent, contract analytical services to the global pharmaceutical and related industries.
Experience of Testing Ninhydrin-Positive Substances using Ph.Eur. Monographs
27 October 2021
The main complication with the analysis of amino acids and related substances is due to their low volatility and inability to form active chromophores when in a solution. This means that GC and HPLC using UV/Vis detection are not applicable and other common HPLC detectors lack the required sensitivity.
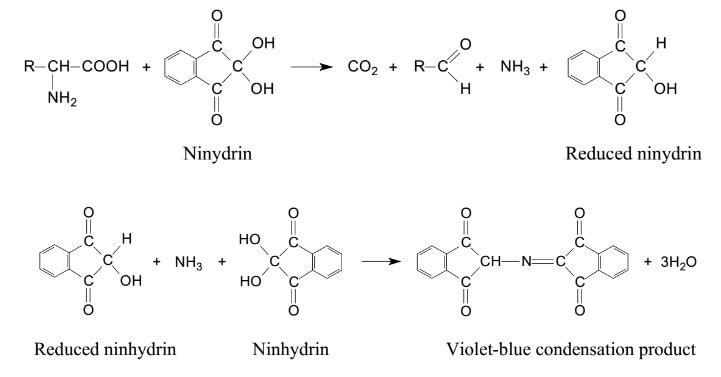
Most free amino acids and ammonium contain a primary amine functional group, which can be derivatised with ninhydrin to form compounds with a chromophore that absorbs electromagnetic radiation at 570nm, resulting in a deep purple colour. Amino acids with a secondary amine functional group can also be derivatised with ninhydrin; these result in chromophores that absorb at 440nm resulting in a yellow colour. Compounds that react in this way are referred to as ninhydrin-positive substances since they give a positive reaction (i.e. colour change) when derivatised with ninhydrin
Historically, the Ph. Eur. Amino Acid monographs took advantage of these reactions to determine the levels of ninhydrin-positive substances present as contaminants in various Amino Acid samples using a Thin Layer Chromatography (TLC) procedure. This involved separating sample components by development of the TLC plate, then visualising the analytes by spraying the plate with ninhydrin which reacts with any ninhydrin-positive substances on the plate to give coloured spots. The major problem with this procedure was that it did not resolve all possible ninhydrin-positive substances. The method was also very subjective since quantitation was based on a simple visual comparison of the relative intensity of sample and standard derived spots. To address the problems with TLC, the Ph. Eur. started revising amino acid monograms in 2014, replacing the TLC requirement with a procedure based on High Performance Liquid Chromatography (HPLC). The new test employed the separation of amino acids on an ion-exchange column with different ionic strength and pH eluents. The resulting stream of separated Amino Acid sample components eluting from the column are mixed with ninhydrin supplied from a secondary pump. The mixed sample and ninhydrin stream is then passed through a heated reaction coil to promote derivatisation of any ninhydrin-positive substances, these can then be detected using a photometer at the wavelengths already specified above. This process is known as ‘post-column ninhydrin derivatisation’. Dedicated instruments capable of post-column ninhydrin derivatisation are commercially available and are known as Amino Acid Analysers (AAA).

With the requirement change from TLC to HPLC post-column ninhydrin derivatisation, Butterworth decided to reconfigure an existing HPLC system with a secondary pump and heated reaction coil to carry out the analysis. However the system lacked sensitivity and, under certain circumstances, by-products from the derivatisation process form precipitates, blocking the tubing and causing high back pressures. These problems increased instrument down time due to the need for maintenance.
To negate these and other problems, and reduce uncertainty with analysis reporting times, Butterworth has purchased and validated a Hitachi LA8080 Amino Acid Analyser. The main differences from the previous ad hoc set-up included an increase in sensitivity. The biggest change was the elimination of tubing blocking and increasing analytical column back pressures. At the end of each individual sample analysis, after all amino acids have eluted, a column regeneration mobile phase is pumped prior to a long period of equilibration with initial analytical mobile phase conditions. Another benefit is that the secondary ninhydrin pump operates in 2 channel mode; a start-up and shutdown program is run with every sequence which involves purging the pump and reaction coil with an ethanol solution to dissolve and remove any particulate build-up.

Butterworth has already verified the new system for the analysis of Arginine, Histidine and Tryptophan, and will verify the method for other less frequently requested amino acids when required by clients. In addition our Projects Department are also currently looking at the same testing in the JP Monograph as the result of a client request.